Overcome process bottlenecks
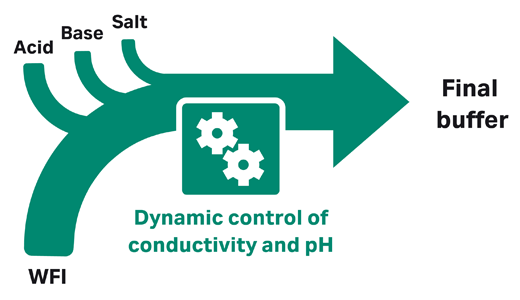
In downstream purification, buffer storage and buffer preparation often take up lots of time and facility space. This can make scale-up difficult, especially when space is limited. Buffer management strategies like inline conditioning (IC), inline dilution, and using ready-made buffers can help.